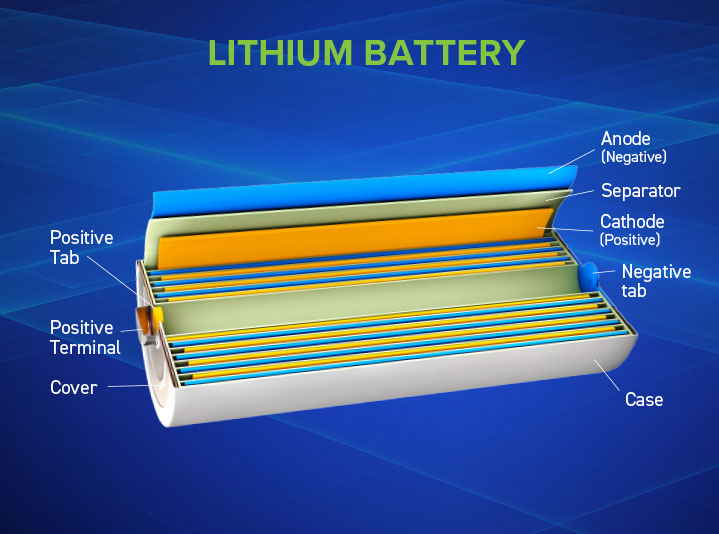
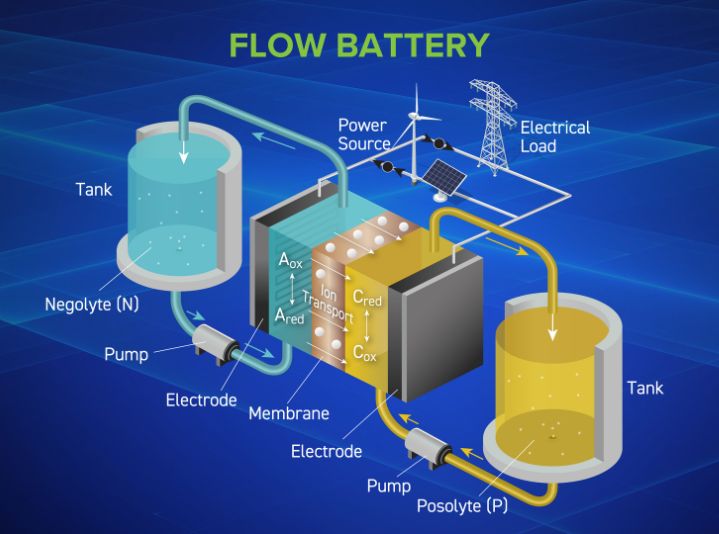
Battery separators are a critical safety and performance component across battery technologies, including lead, flow, and lithium-ion. Though often overlooked, the separator plays a central role in enabling chemical reactions to occur safely and efficiently across battery chemistries – supporting reliable operations from automotive starting batteries to high-capacity industrial systems.
About Lead Batteries
Today’s innovative lead batteries are to a cleaner, greener future and provides 50% of the world’s rechargeable power.