The BCI Battery Poster Research Showcase is open to undergraduate, graduate, and postdoctoral level researchers and assistants to share their latest work during the BCI Convention + Power Mart Expo. Learn more about the showcase.

2025 – Tim Fister, Argonne National Laboratory
Acoustic Characterization of Lead Acid Batteries
Local variation in electrode and electrolyte composition contributes to large-scale changes in overall utilization and cycle life of lead acid batteries. Here, we outline the potential for ultrasonic characterization of lead acid batteries to diagnose local changes in state of charge (SOC) and state of health (SOH). Using 2V cells constructed with commercial battery plates, we show that the amplitude and time-of-flight of ultrasonic signals is highly sensitive to changes in the bulk modulus and density of lead battery materials and acid specific gravity during cycling. Using high-rate partial state of charge cycling, we also show that these changes can be used to track the onset of sulfation on the negative electrode. This nondestructive, analytical tool can provide crucial feedback for emerging battery management systems and could potentially be used for quality control measures during manufacturing.

2025 – Cailin Buchanan, Argonne National Laboratory
Accelerating Redox Flow Battery Development Through an Autonomous Flow Battery Cycler Station
Redox flow batteries (RFBs) are a promising technology for long duration energy storage (LDES) because of their theoretically long cycle life and scalability. Although the state-of-the-art RFB is vanadium-based, it is not economical due to vanadium’s price volatility and low voltage. Thus, RFBs with alternative electrolytes must be explored. Organic RFBs are attractive due to their low-cost, earth-abundant materials, however, many systems suffer from poor stability and low energy density. Given the vast parameter space for RFB electrolytes, including active materials and supporting electrolytes, the pace of research urgently needs to be quickened to identify systems that are stable and have high energy densities. To accelerate the development of RFBs, we have built an automated flow battery cycler station that can explore a large parameter space and leverage artificial intelligence (AI). The automated platform contains sixteen electrochemical testing channels, each comprised of four peristaltic pumping channels and four selector valves that deliver electrolyte to the flow cell. One software controls both the electrolyte delivery system and the electrochemical testing, resulting in complete automation and closed-loop capabilities. To test the automated station, we collected electrochemistry results for commonly proposed redox active materials like hydroxy-TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxy) and supporting salts like lithium bis(trifluoromethanesulfonyl) imide (LiTFSI). The automated system was used to explore a wide range of supporting salt concentrations without human intervention, underscoring the utility of the cycler station and its potential for use in an autonomous discovery platform to accelerate LDES technology development even further when coupled with machine learning optimization algorithms.

2024 – Cailin Buchanan, Argonne National Laboratory
Investigation of organic expander molecules to advance understanding of structure-function relationships in lead acid batteries
Expander molecules like Vanisperse A are added to the negative electrode pastes used in lead acid (PbA) batteries to promote high surface area and favorable discharge performance. Despite these advantages, expander molecules typically inhibit charging rates, limiting the use of PbAs in advanced applications that require repeated deep discharge/charge cycling. A deeper understanding of the atomic-level mechanisms that control additive-lead species interactions is necessary to optimize expander molecules for both discharge and charge performance. A collaborative project between government, academia, and industry has screened over one hundred model expander molecules (MEMs) using cyclic voltammetry, density functional theory, and various spectroscopy methods to characterize their chemical and electrochemical stability and performance properties. The MEMs are categorized by their lignin structural motifs and the presence of functional groups, e.g., sulfate, sulfonate, and carboxylate, with the goal of establishing structure-function relationships. Discharge (DEF) and charge (CEF) enhancement factors were established as the metrics for electrochemical performance relative to sulfuric acid without expanders as the baseline. Four categories of expander molecules were defined based on their DEF and CEF values: traditional, e.g., Van A, inhibitors, enhancers, and rheology modifiers. The set of materials evaluated to date demonstrates that expander molecules that can enhance both the discharge and charge performance are possible and do exist. On the other hand, the inhibitor class may lead to a deeper understanding of the expander degradation processes and their impact on cycle life. These results help us identify the design rules for expander molecules targeted to advanced PbA applications.

2024 – Madhu Chennapuram, The University of Toledo
Design, synthesis and structural evaluation of model expander molecules for advanced lead-acid battery storage applications
Lead acid batteries provide a day-to-day reliable energy storage application in various fields like automotive, standby power, renewable energy, telecommunication, industrial and robotics. 1 In addition to the electrochemically active lead species, these batteries contain a number of additives that improve performance and cycle life. Lignosulfonates (LS) are organic biopolymers that are used as additives in a variety of applications, including the production of lead acid batteries (Figure 1) 2. In lead acid batteries, LS serve as organic expanders to improve the performance of the battery’s storage capacity and extending its service life, as well as by acting as a wetting agents and improving the conductivity of the electrolyte, which tends to improve battery efficiency. 3 To be able to understand the interaction of specific functional groups with lead species in detail, small molecules that mimic portions of LS can be used as model expander molecules (MEMs). In this study, we are designing and synthesizing lignosulfonate-based MEMs. A series of MEMs were prepared from different synthetic methods (Scheme 1). The MEMs’ stability under conditions relevant for battery applications was investigated by cyclic voltammetry in 5 M H2SO4. Additionally, the interaction of the MEMs with Pb2+ and their stability at elevated temperature and in 5 M sulfuric acid was studied by spectroscopic and diffraction techniques.
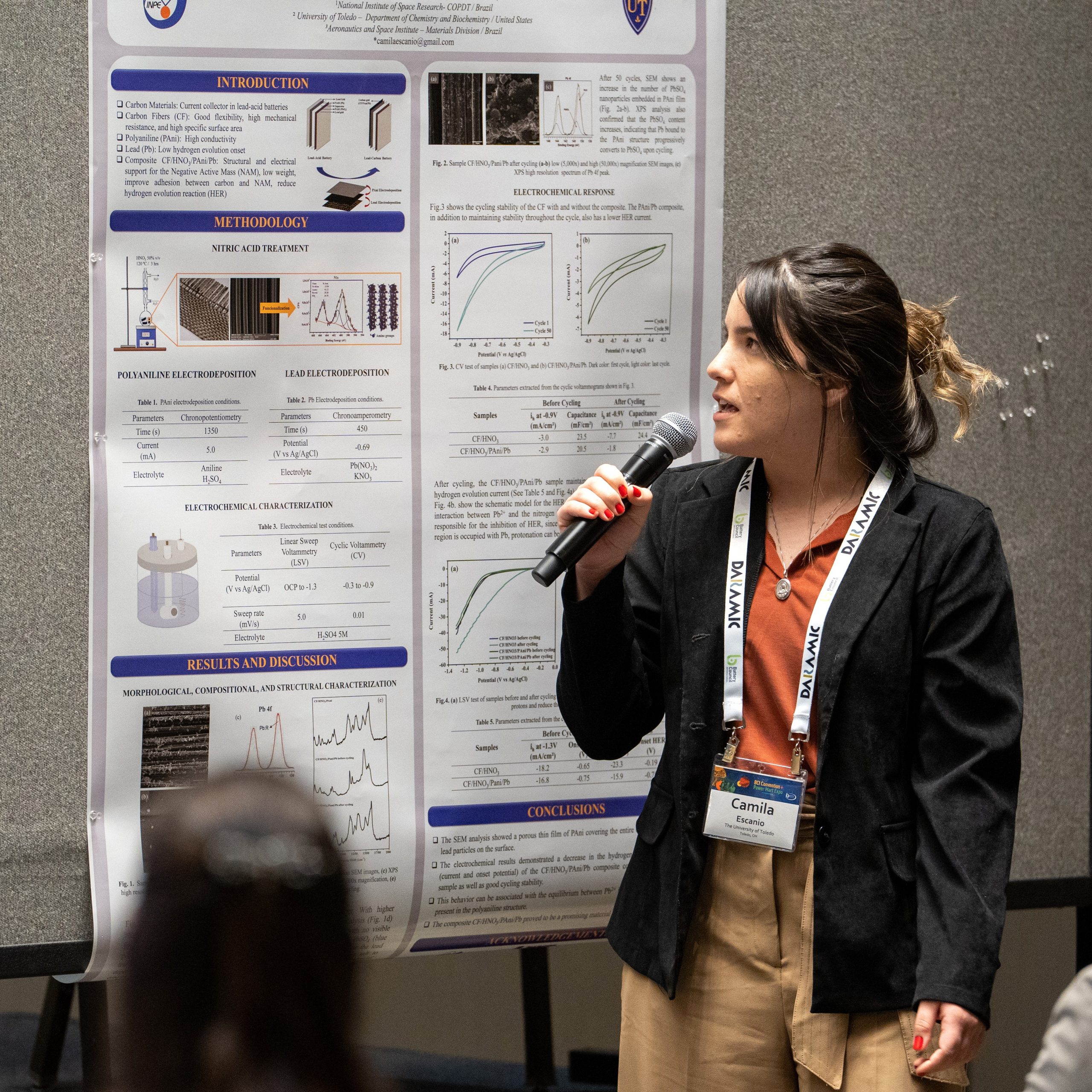
2023 – Camila Alves Escanio, The University of Toledo/National Institute for Space Research
Carbon Fiber/Polyaniline/Lead Composite With Low Hydrogen Evolution Activity With Potential Application in Lead Acid Battery
A composite made of Carbon fiber (CF), Polyaniline (PAni), and lead (Pb), with low hydrogen evolution activity, was developed and studied as a potential material to be used in lead acid batteries. First, the CF was subjected to acid treatment using 50% v/v HNO3 to remove the sizing and add nitrogen functional groups on the carbon surface (CF/HNO3). The second step, PAni electrodeposition (CF/HNO3/PAni), was performed through chronopotentiometry by applying a constant current of 5 mA for 1350 s. Next, the lead electrodeposition was carried out by chronoamperometry by applying the lead reduction potential (-0.69 V vs Ag/AgCl) for 450 s. Finally, the composite CF/HNO3/PAni/Pb was dried in a vacuum oven at 60 °C for 1 h. Morphological and structural analyses were performed using Scanning Electron Microscopy (SEM) and Raman Spectroscopy. In addition, the electrochemical behavior, such as the hydrogen evolution phenomenon and cycling stability, was evaluated by Linear Sweep Voltammetry and Cyclic Voltammetry, respectively. The SEM analysis showed a porous thin film of PAni covering the entire carbon fiber with some lead particles on the surface. Furthermore, the electrochemical results demonstrated a decrease in the hydrogen evolution phenomenon (current and onset potential) of the CF/HNO3/PAni/Pb composite compared to the CF/HNO3 sample as well as good cycling stability. Acknowledgment: The authors are grateful to FAPESP (process nº 2022/10542-4 and nº 2019/08473-1) for the financial support.

2023 -Ravi Shankar Raju Madiraju, Hammond Group
Brunauer-Emmett-Teller (Bet) Analysis for the Surface Area of Ingredients in Lead Acid Battery
As particles get smaller, their surface area to volume ratio rises. The multi-molecular adsorption technique “BET”, to determine the surface area was invented by Stephen Brunauer, Paul Hugh Emmett, and Edward Teller in 1938. Brunauer, Emmett, and Teller expanded Langmuir’s (1918) adsorption to several molecular layers to understand how adsorption data affects material attributes such as total surface area, pore-size distribution, micro-pore analysis, and porosity. BET method is used for both macro and nano materials and is a common technique used to classify raw materials and active materials in the battery industry. This study mainly concentrates on finding the correlation coefficient for each ingredient of a lead-acid battery paste mix against BET surface area values. This is done by developing a master data matrix that collects weight percentages of the ingredients in a lead acid battery paste mix with respect to lead-oxide content. The BET surface area results of the dry cured battery active material are then compared to the weight percentages of each ingredient to determine that ingredient’s effect on surface area. Correlation coefficients measure linear relationships between variables. -1 to 1 are its values. According to a correlation value of -1, one series rises while the other falls, and vice versa. 1 is a perfect positive connection. Zero correlation means no linear relationship.
The BCI Battery Poster Research Showcase is open to undergraduate, graduate, and postdoctoral level researchers and assistants to share their latest work during the BCI Convention + Power Mart Expo each year.
Sustaining long-term success in our industry requires more than innovation. It requires strong financial stewardship, a values-driven culture, and a commitment to delivering for our customers and communities.


