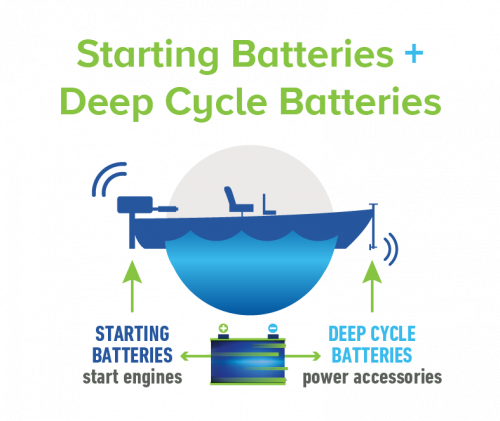
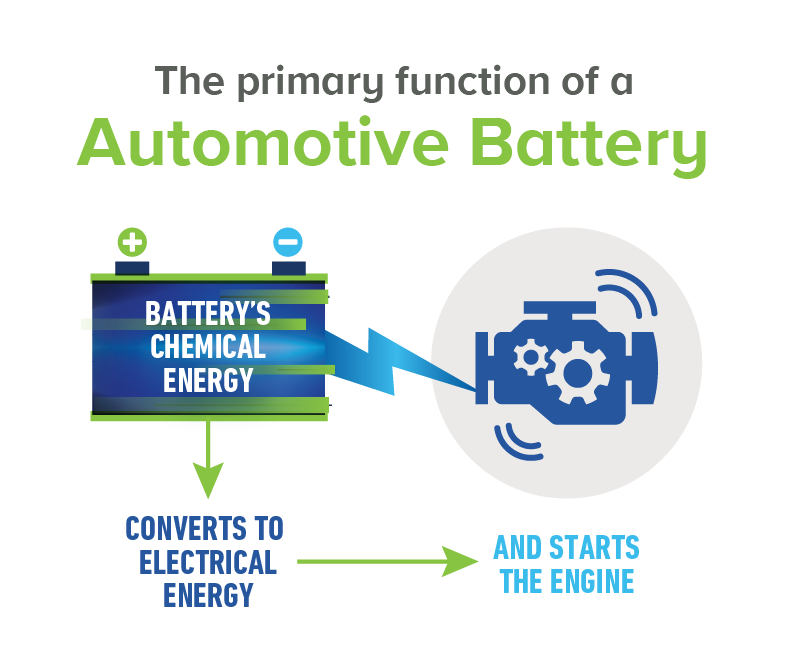
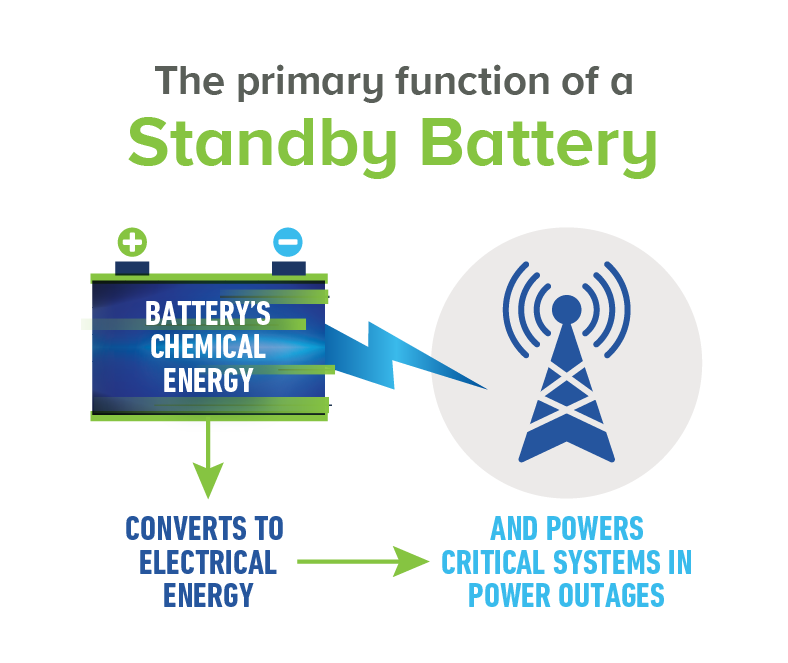
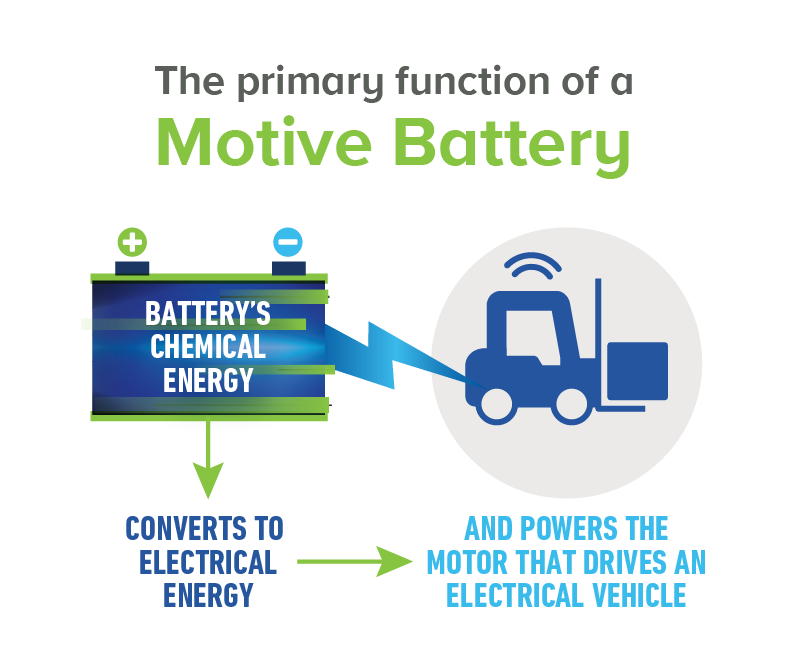
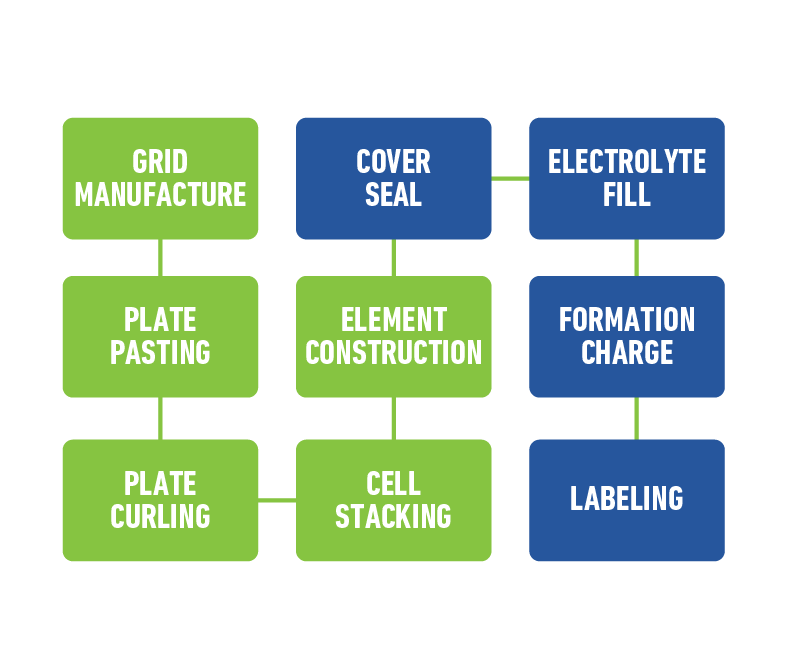
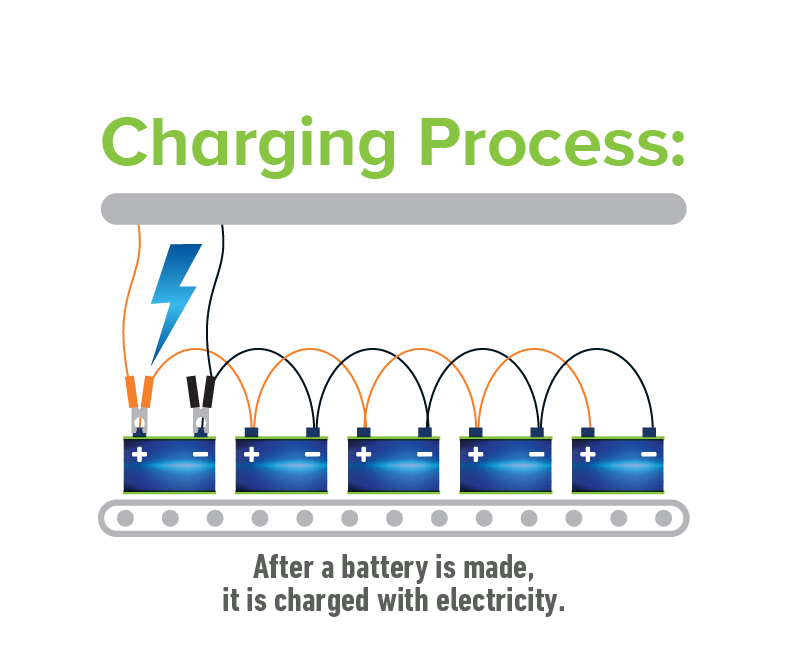
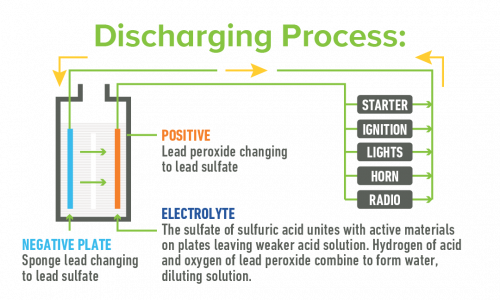
Today’s innovative lead acid batteries are key to a cleaner, greener future and provide 50% of the world’s rechargeable power. They’re also the most environmentally sustainable battery technology and a stellar example of a circular economy.
A Model of Sustainability and Circularity
Today’s innovative lead batteries are key to a cleaner, greener future. They’re also the most environmentally sustainable battery...